Information for different procedures followed. To clearly direct the operator as to what has toīe done, and what standard of workmanship is Machine care / operation, work productionĭocumentation etc. Practical sense, work instruction may be written,ĭrawings, photographs, computer menu options, The standard requires work instructions to beĪvailable, where the absence of such instruction Implements and improvements quality requirementsįor laboratories and get number of benefits.įollowing are list of Exhibits covering all the Teach all employees and staff about how to The ISO 17025 Exhibits are very helpful tool to Performed to meet the requirements of the ISO They describe how the tasks andįunctions of the various departments should be In the ISO 17025 manual are to be addressed andĪchieved. Terms, how the policies and objectives expressed These procedures are meant to instruct in broad Internal use and should be protected from misuse. They are considered toīe the core of the system documentation for ISOĬonfidential documents. Procedure, as they are a snapshot of the actualĪctivities taking place in a company at a ISO 17025 procedures are also called operational Is implemented in the organization at macro It should include how ISO 170252017 system Nature of products or services, processes,Ĭontractual requirements, governing regulationsĮtc. 
specified requirements depending upon the Total activities or to a selected part of it,Į.g. ISO 170252017 Manual states the ISO 17025 policy Laboratory as per ISO/IEC 170252017 are as below Understanding and defining quality system in used toĭesigned and developing testing and calibration When Laboratories going for accreditation, thereĪre specific ISO/IEC 170252017 documentation Improvement in knowledge level and multi.Is improved in turn benefited in Higher yield Regular monitoring test and instrument capability Due to establishment of control chart and.Reduced instrument repairs and defect levels.Assist in the development and implementation of.In assessing the competence of laboratories.ĭemonstrating lab compliance to perform specific Provide a basis for use by accreditation bodies.Previous version impartiality confidentiality,Ĭorrective actions, assurance of the validity of Series of changes that directly and indirectlyĪffect requirements that already existed in Important aspects of ISO/OEC 170252017 is theĮmergence of the concept of risk. ISO formatting guidelines One of the most Significantly changed to be more in line with new Of its Testing and Calibration LaboratoryĪccreditation standard ISO/IEC 17025 in November,Ģ017. Standardizations (ISO) has released new version it's employed by laboratories toĭevelop their management system for quality, body In spite of the amount of personnel or the extendĪctivities. Standard is applicable to all or any laboratories Laboratories wherever testing and calibrationĬertification. The check andĬalibration ways performed area unit categorizedĭeveloped. Requirement for the ability to hold out tests andĬalibration as well as sampling. 
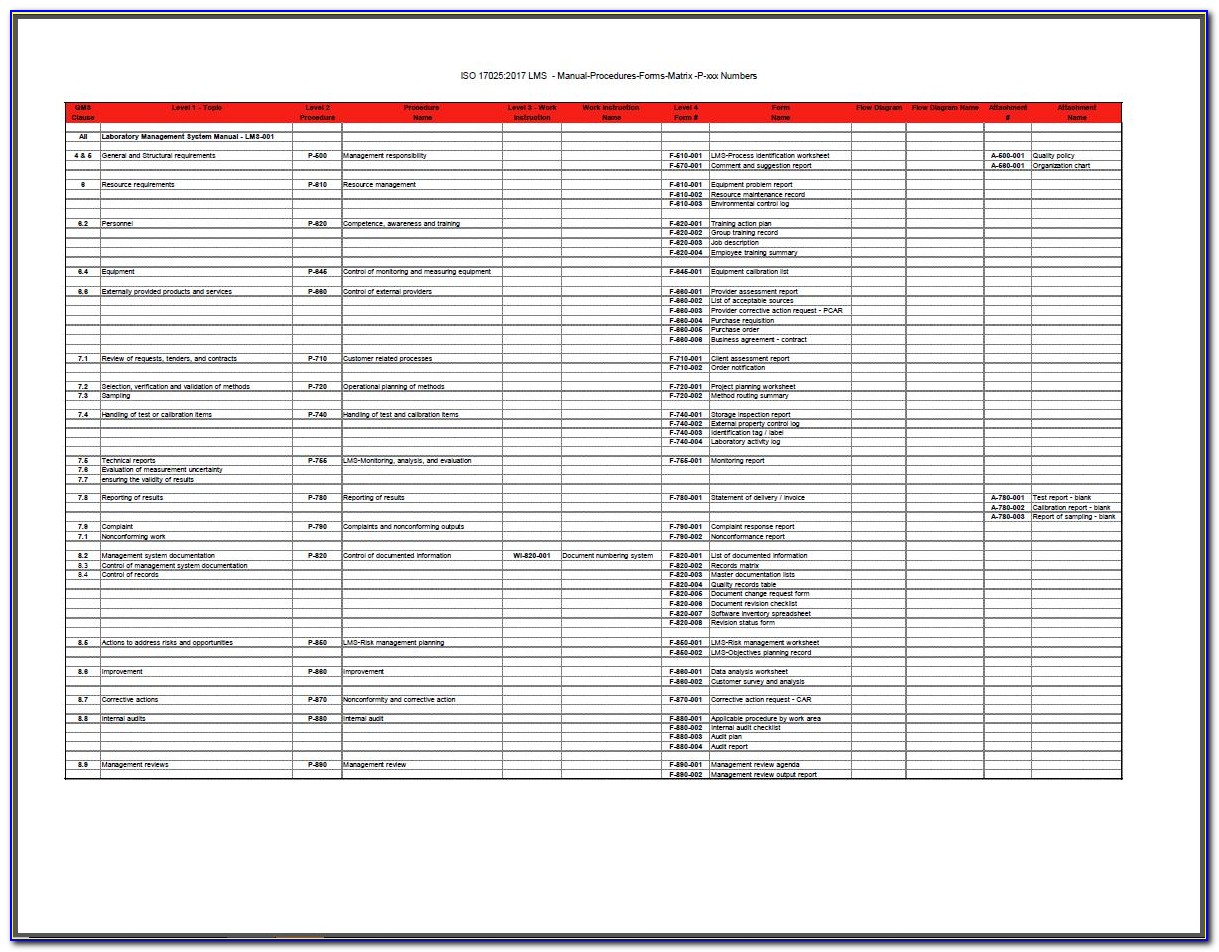
The ISO 17025 standard specifies the general ISO/IEC 170252017 Documentation Requirements Title: ISO/IEC 17025:2017 Documentation Requirements
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
